The Evolving CML Market – which drug to give when?
One of the enduring legacies from the development of imatinib (Gleevec®/Glivec®) for the treatment of chronic myeloid leukemia (CML) is the long-term survival data from the IRIS (International Randomized Interferon versus STI571) trial that enrolled 1106 patients between June 2000 and January 2001.
 Hagop Kantarjian, M.D. Professor and Chair, Department of Leukemia at The University of Texas MD Anderson Cancer Center in Houston told the 2012 Chemotherapy Foundation Symposium (also known as the Greenspan Meeting in honor of the late Ezra Greenspan, M.D.) that:
Hagop Kantarjian, M.D. Professor and Chair, Department of Leukemia at The University of Texas MD Anderson Cancer Center in Houston told the 2012 Chemotherapy Foundation Symposium (also known as the Greenspan Meeting in honor of the late Ezra Greenspan, M.D.) that:
the 10 year survival rate is 85% in patients treated with imatinib, and that this rises to 90% if you exclude deaths not related to CML.
No second-generation tyrosine kinase inhibitor (e.g. nilotinib, dasatinib, bosutinib) has yet to show a superior long-term survival benefit to imatinib. Dr Kantarjian noted that “whatever comes as a new treatment in the frontline therapy has to be able to beat that time, of a 10 year survival, if there is a big cost difference.”
The IRIS trial survival data for imatinib remains the gold standard by which other tyrosine kinase inhibitors will be judged.
In other words, notwithstanding the myriad of published CML data that shows second generation TKIs offer a deeper or more rapid molecular response, there’s still no data that shows you will actually live longer if you take any of them instead of imatinib.
That’s not to say there are no benefits to the newer second generation TKIs.
This content is restricted to subscribers
 However, it is interesting that we have not seen Novartis publish an update to the IRIS trial for a while. Instead, faced with imatinib going off patent in 2014, Dr Kantarjian noted that the company has raised the price of imatinib and destroyed the price differential to nilotinib to encourage more nilotinib usage. It’s hard to compete against your own successful product!
However, it is interesting that we have not seen Novartis publish an update to the IRIS trial for a while. Instead, faced with imatinib going off patent in 2014, Dr Kantarjian noted that the company has raised the price of imatinib and destroyed the price differential to nilotinib to encourage more nilotinib usage. It’s hard to compete against your own successful product!
In the short-term, this marketing strategy may promote the use of nilotinib instead of imatinib. The reality, however, is that we can expect generic imatinib to be widely used frontline once available, where cost is an issue, whether it be for out of pocket patient co-pays, or in countries where healthcare reimbursement is government funded. It will be interesting to see whether this will put some pressure on companies to lower the price of second-generation TKI’s.
In terms of current treatments for CML, Dr Kantarjian told a large audience at the Greenspan Meeting that the choice of FDA approved TKI therapy for CML in frontline is imatinib 400 mg daily, nilotinb 300 mg BID or dasatinib 100 mg daily. In second or third line therapy, the choice of TKI options is nilotinib, dasatinib, bosutinib or ponatinib (pending FDA approval in 2013).
Choice of TKI in CML
Dr Kantajarian discussed in detail the factors that clinicians need to consider when choosing a TKI for patients with CML. These include:
- Efficacy – CGCR, MMR, CMR, EFS, survival
- Toxicities
- Disease status – frontline, salvage, tranformation
- Salvage – Mutation status
- Comorbidities: COPD, CHF, diabetes, pancreatitis
- Cost
It’s beyond the scope of a blog post to go into detail on these. However, when it comes to efficacy, it is important to look at outcomes.
Dr Kantarjian told the audience that with second generation TKIs “so far there is no survival benefit” over imatinib and that “many patients who fail imatinib can be salvaged effectively with second TKIs.”
Important Response Categories in CML
If you are looking for an improvement in survival, you need to achieve a complete cytogenetic response, you do not have to have a major molecular response, said Kantarjian.
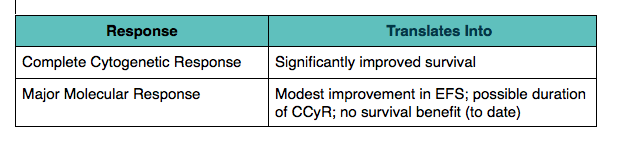
Table modified from Hagop Kantarjian, M.D. presentation at 2012 Chemotherapy Foundation Symposium
Dr Kantarjian noted that in a survey of 507 U.S. community oncologists: 72% had Major Molecular Response (MMR) as the desired treatment outcome with only 17% seeking a Complete Cytogenetic Response (CGCR).
The Golden Rule in CML Monitoring
Dr Kantarjian shared with the Chemotherapy Foundation Symposium audience some of his golden rules in CML monitoring, one of which was:
“Do not discard a TKI unless there is a loss of CGCR (not MMR) at the maximum tolerated adjusted dose that does not cause grade 3-4 or chronic grade 2 (affecting QOL) toxicities.”
He went on to advise that when analyzing mutations in CML:
- If CG or hematologic relapse occur, mutations studies help
- No role for mutation studies pre-Rx or in imatinib responding patients
As for the choice of TKI at the MD Anderson Cancer Center, Dr Kantarjian told the audience that they currently use investigational ponatinib in both frontline and salvage settings. He noted that as an investigational drug it was free to the institution and patients.
The CML market remains one that will undergo further evolution over the next few years as new drugs such as ponatinib come to market and existing drugs such as imatinib go off patent.
It will be interesting to watch the market dynamics and whether the clinical benefits of the second-generation TKI’s justify their significantly higher cost over generic imatinib.