Urologist Castrates Media over Reporting of PIVOT Prostate Cancer Trial
Dr Benjamin J. Davies, an academic urologist at the University of Pittsburgh today castrated the media over their coverage of the Prostate Cancer Intervention versus Observation Trial (PIVOT).

In an article titled “Prostate Cancer: Lessons from PIVOT lost in media hype” published in the News and Views section of Nature Reviews Urology, Dr Davies states, “we must be careful to ensure the less-newsworthy facts and limitations of high-profile trials, such as PIVOT, are not lost in the media hype.”
Davies goes on to say,
“an odious meme is circulating in the medical media, suggesting that prostate cancer is universally diagnosed, that PSA screening causes more harm than help, and that urologists should disregard basic epidemiologic data.”
Strong words perhaps, but those who follow Davies on twitter (@daviesbj) will know that he does not mince words and is not lost for an opinion.
However, in writing for a publication such as Nature Reviews Urology, which is probably not on the reading list of the private practice urologist or member of the mass media, he is preaching to the converted, namely academic-orientated physicians like Davies himself.
All clinical trials have their limitations, and Davies makes valid points that the PIVOT trial has a number of noticeable weaknesses. Attention was also drawn to this in the accompanying editorial when the data was recently published in the New England Journal of Medicine. I encourage you to read his review.
I reported the presentations of the PIVOT data from the plenary sessions at the 2011 annual meeting of the American Urological Association (AUA) and the 2012 congress of the European Association of Urology (EAU) on this blog and do take exception to Davies’ implied assertion that ALL the media coverage of the PIVOT trial was “hype.”
Experienced Healthcare journalists such as Scott Hensley (@scotthensley) provided fair and evenly balanced coverage on NPR Shots, for example.
If the media coverage of the PIVOT trial data was not as balanced or did not contain the message that Davies wanted to hear, then rather than shoot the media messenger the urology community should ask themselves why they did not obtain it?
Interestingly, at AUA 2011 and EAU 2012 there were no press conferences on the PIVOT trial data, yet it was an important topic and a plenary presentation. Press conferences allow the media to ask questions of a panel of speakers and the opportunity to gain a variety of perspectives. Why did the leading urologists who organize these major medical congresses not provide this access?
It is the responsibility of the urology community to reach out and educate the media if you think we don’t understand the nuances of the data.
Davies singles out the PIVOT trial for critical review, but in so doing he touches upon the wider issue of the lack of quality clinical trial data to support treatment and practice in urology. It is for this reason that those clinical trials that are published, whatever their limitations, have disproportionate impact.
As I wrote from EAU 2012, why is there no level 1 evidence-based medicine that shows the benefits of robot assisted radical prostatectomy? Are academic physicians unable to do high quality and robust clinical trials that justify their practice?
In his article, Davies goes beyond criticizing the PIVOT trial to castigating the media over their coverage of PSA screening, for which he is an ardent proponent.
Unfortunately, he ignores the reality that mass media don’t generate the data, they only report what organizations such as the United States Preventative Services Task Force (USPSTF) recommend. If academic urologists believe the USPSTF got it wrong, then the failure is theirs in their inability to generate compelling data or influence the recommendations.
Finally, when Davies says, “no doubt urologists have not helped themselves by overscreening and overtreating” he touches on what I believe is the underlying cause of much of the problem associated with PSA screening.
Academic urologists need to educate their community colleagues. Influencing everyday practice and treatment decisions will do more to help patients in the long run than being critical of the media, however justified that may be in some cases.


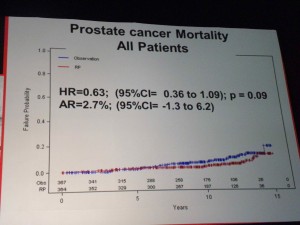
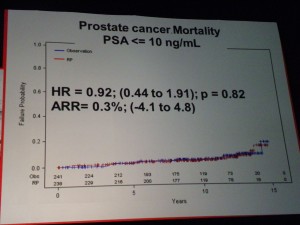 However in those patients with PSA > 10ng/ml there was a significant difference in mortality (p=0.03 HR=0.36) suggesting that in the small sub-group of patients with high PSA, RP did provide a benefit over “watchful waiting.” PSA measurements may therefore help in the treatment and ongoing management of patients.
However in those patients with PSA > 10ng/ml there was a significant difference in mortality (p=0.03 HR=0.36) suggesting that in the small sub-group of patients with high PSA, RP did provide a benefit over “watchful waiting.” PSA measurements may therefore help in the treatment and ongoing management of patients.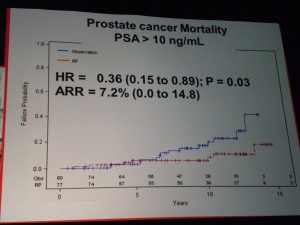 If one of the challenges of PSA screening is the issue of the harms from treatment, then the PIVOT data that showed no difference in mortality through watchful waiting over radical prostatectomy (RP) except for high risk patients is important.
If one of the challenges of PSA screening is the issue of the harms from treatment, then the PIVOT data that showed no difference in mortality through watchful waiting over radical prostatectomy (RP) except for high risk patients is important. The market for prostate cancer therapies is set to expand from $1 billion currently to $5 billion by 2015, according to analysts reported by
The market for prostate cancer therapies is set to expand from $1 billion currently to $5 billion by 2015, according to analysts reported by