The patient advocacy session at the recent 16th Congress of the European Hematology Association in London focused on adherence to cancer treatments, and was filled to capacity, with the many attendees having to watch it from an overflow area.
Dr David Marin, Reader in Onco-Haematology at Imperial College, London presented research published last year in the Journal of Clinical Oncology that dramatically demonstrated how adherence to chronic myeloid leukemia (CML) therapy is the critical factor for achieving molecular responses.
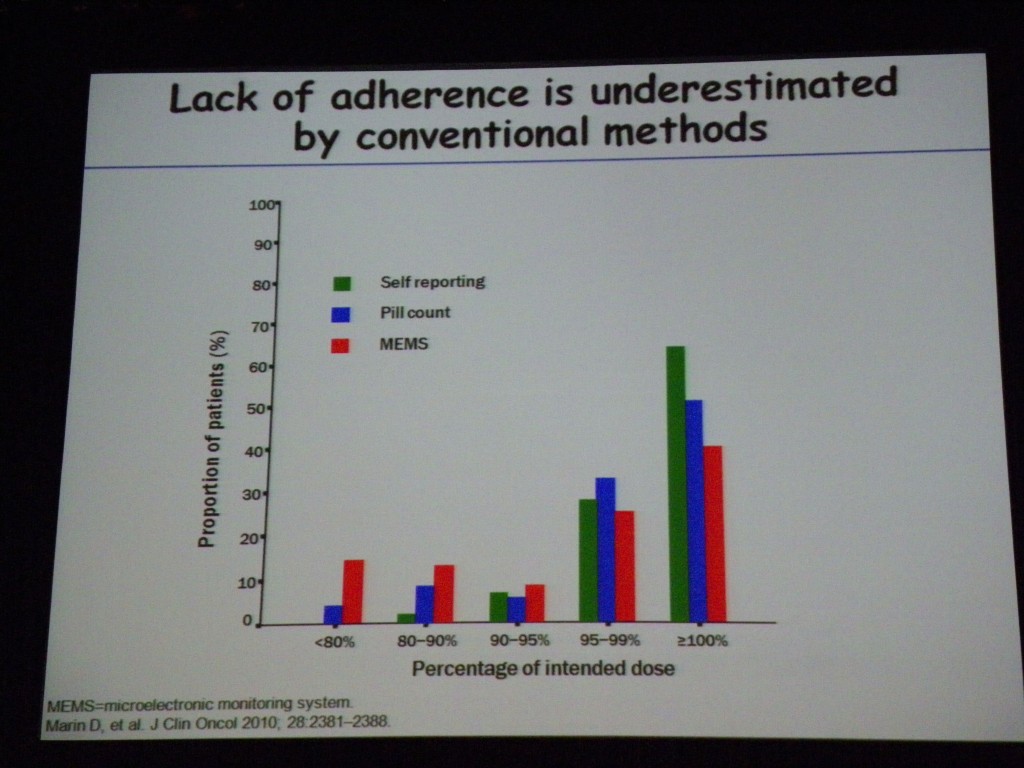
In a study of 87 CML patients taking imatinib (Glivec®/Gleevec®) for a median period of 91 days, Dr Marin showed that no major molecular response (MMR) was observed when adherence was ≤ 80% and no complete molecular responses (CMR) were observed when adherence was ≤ 90%. The graphical figure that he presented from his paper, dramatically shows how missing only a few doses of drug can have a major impact on outcome:

Although the work by Marin and colleagues at the Hammersmith Hospital was undertaken with CML patients taking imatinib, the paper notes that adherence problems
“may apply equally to patients receiving second-generation tyrosine kinase inhibitors.”
Imatinib is the only TKI approved in the UK, thus that’s the only one available for studies there to date.
What made this data so compelling was the study rationale that used an electronic pill container, the medical event monitoring system (MEMS™) from the Aardex Group. This product contains a microchip that records the date and time it is opened.
Dr Marin’s study showed that “lack of adherence is underestimated by conventional methods.” Self-reporting of adherence and pill counts are inaccurate compared to electronic data capture using MEMS (study subjects were unaware of the micro-chip in the pill bottle).
When psychologists at the Hammersmith Hospital subsequently interviewed patients who missed doses of drug, they found intentional and non-intentional adherence reasons.
A few excepts of patient quotes from Dr Marin’s presentation:
Intentional non-adherence:
“Oh I can’t be bothered tonight, it’s not going to kill me [to miss a dose]”
“I thought there was no way I was going [on holiday] and being tired.”
Unintentional non-adherence:
“And sometimes you just forget”
“[the pharmacy] had no medication for me, so I went for nearly a week with no medication.”
Other speakers in the excellent patient advocacy session chaired by Jana Pelouchová (European Cancer Patient Coalition, Czech Republic) and Jan Geissler (CML Advocates Network, Germany) included Giora Sharf (Israeli CML patient’s Organization and CML Advocates Network, Israel) and Professor Rudolf Schoberberger (Medical University of Vienna, Austria).
Professor Schoberberger focused on the impact of drug packaging on compliance, particularly in elderly patients, and presented compelling research on how “child-proof” equals “age-proof.” Sally Church in her video blog from EHA also discusses the patient advocacy session and how pharma/biotech companies could improve drug packaging.
The issue of adherence is a personal choice that every patient taking a chronic therapy makes. However, as Sally notes on Pharma Strategy Blog more patient and physician education is needed so that patients know there may be dramatic consequences from missing only a few doses per month.
Not only may adherence have a major impact on patient outcome, but as one questioner from France pointed out at the end of the EHA patient advocacy session, “for a statistician it is a nightmare.” Poor adherence in clinical trials “means that the true effect of a drug is not well known. Efficacy may be under-estimated if adherence is low.”
More monitoring of adherence in clinical trials through the use of MEMS technology may, therefore, be necessary to ensure that clinical trial data shows the true efficacy and adverse event profile of a drug.
I hope that the European Hematology Association (EHA) will make a webcast of this informative patient advocacy session publicly available online as it raised issues of considerable importance to patients, physicians and biotech/pharma companies alike.
 Marin, D., Bazeos, A., Mahon, F., Eliasson, L., Milojkovic, D., Bua, M., Apperley, J., Szydlo, R., Desai, R., Kozlowski, K., Paliompeis, C., Latham, V., Foroni, L., Molimard, M., Reid, A., Rezvani, K., de Lavallade, H., Guallar, C., Goldman, J., & Khorashad, J. (2010). Adherence Is the Critical Factor for Achieving Molecular Responses in Patients With Chronic Myeloid Leukemia Who Achieve Complete Cytogenetic Responses on Imatinib Journal of Clinical Oncology, 28 (14), 2381-2388 DOI: 10.1200/JCO.2009.26.3087
Marin, D., Bazeos, A., Mahon, F., Eliasson, L., Milojkovic, D., Bua, M., Apperley, J., Szydlo, R., Desai, R., Kozlowski, K., Paliompeis, C., Latham, V., Foroni, L., Molimard, M., Reid, A., Rezvani, K., de Lavallade, H., Guallar, C., Goldman, J., & Khorashad, J. (2010). Adherence Is the Critical Factor for Achieving Molecular Responses in Patients With Chronic Myeloid Leukemia Who Achieve Complete Cytogenetic Responses on Imatinib Journal of Clinical Oncology, 28 (14), 2381-2388 DOI: 10.1200/JCO.2009.26.3087