The dirty little secret biotechs don’t tell you
 Do not feed the alligators is a common sign seen in the hammock areas of Florida where brackish fresh water abounds.
Do not feed the alligators is a common sign seen in the hammock areas of Florida where brackish fresh water abounds.
It has always amazed me people might even consider getting close enough to see them since common sense would tell you to beat a hasty and instant retreat from those big toothy maws!
Similarly in biotechland sometimes there are warning signs aplenty and yet people still ignore them, preferring to focus on the good.
Today’s story is a cautionary one where, much like with those “Do not feed the alligators” signs, researchers and investors might be wiser to heed certain warnings rather than focusing solely on what initially appears to be positive data.
Ignore the warning signs at your peril…
To continue reading our latest highlights on oncology new product development including commentary and analysis BSB subscribers can log-in or you can click to access the content.
This content is restricted to subscribers

 Science drives oncology new product development and the AACR special conferences are always a good place to look for insights into where the field is both emerging and also going in the future.
Science drives oncology new product development and the AACR special conferences are always a good place to look for insights into where the field is both emerging and also going in the future.

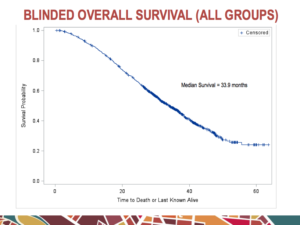
 At the recent ASCO 2016 Genitourinary Cancers Symposium (ASCO GU) that took place in San Francisco the week before the JP Morgan Healthcare Conference (JPM), one of the noteworthy presentations was on a novel target for men with advanced prostate cancer.
At the recent ASCO 2016 Genitourinary Cancers Symposium (ASCO GU) that took place in San Francisco the week before the JP Morgan Healthcare Conference (JPM), one of the noteworthy presentations was on a novel target for men with advanced prostate cancer.